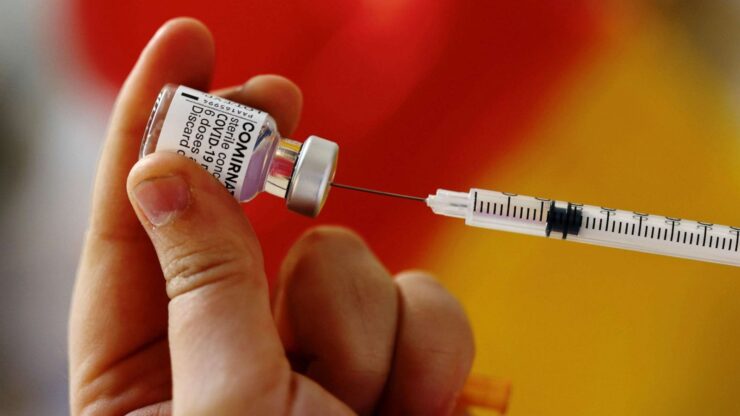
Pfizer and its vaccine partner BioNTech have started an application to request the Food and Drug Administration’s approval for its COVID-19 vaccine.
Pfizer is the first coronavirus vaccine maker in the U.S. to request full approval. Like Pfizer, the Moderna and Johnson & Johnson vaccines had been previously cleared for use under the agency’s emergency use authorization — a less rigorous approval method to aid a swifter response to the pandemic.
An FDA approval for a vaccine means the agency has decided that its benefits outweigh the known risks following a review of the manufacturer’s testing results.
https://opoyi.com/canelo-alvarez-vs-billy-joe-saunders-fight-live-streaming-online
https://opoyi.com/iSljgbZ9GPZy
https://opoyi.com/watch-canelo-vs-saunders-live-stream-boxing-full-fight-online-free
https://opoyi.com/canelo-lvarez-vs-billy-joe-saunders-fight-live-stream-boxing-08052021
https://opoyi.com/watchcanelo-vs-saunders-free-live-stream080521-full-fight-boxing-online
https://opoyi.com/canelo-alvarez-vs-saunders-live-stream-free-boxing-fight
https://opoyi.com/canelo-vs-saunders-results-live-stream-updates-of-the-undercard-main-card-online-may-8-saturday
If granted, Pfizer’s full stamp of approval would only apply to the vaccine for people who are 16 and older. Meanwhile, the vaccine maker is seeking emergency use authorization for its COVID-19 vaccine to include children between 12 and 15.
“We look forward to working with the FDA to complete this rolling submission and support their review, with the goal of securing full regulatory approval of the vaccine in the coming months,” Pfizer CEO Albert Bourla said in a press release Friday.
BioNTech’s CEO, Dr. Ugur Sahin, called the submission of the application for FDA approval “an important cornerstone of achieving long-term herd immunity and containing COVID-19 in the future.”
“We are pleased to work with U.S. regulators to seek approval of our COVID-19 vaccine based on our pivotal Phase 3 trial and follow-up data.”
In that final phase of clinical trials, Pfizer found that its vaccine was 95% effective against COVID-19, the highest efficacy among vaccines authorized for emergency use in the United States.
For the FDA to grant approval, Pfizer-BioNTech will have to comply with a key requirement: providing follow-up data six months after vaccinations.